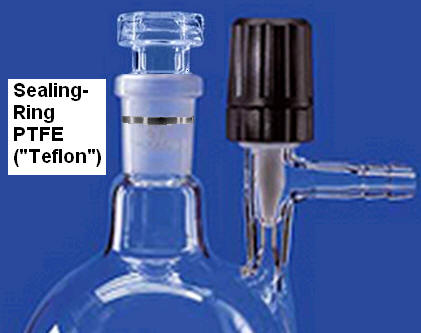
Glindemann PTFE Sealing Rings
Distributors - See table of catalogue numbers of major distributors. For example:
Sigma-Aldrich (in 200
countries,
search their shop for
"glindemann")
Lab Unlimited Carl Stuart
Group
www.labunlimited.com
(worldwide including USA, search their shop for "glindemann")
Carl Roth in more than 100 countries (not in North America).
Please contact Dr. Glindemann at
dglinde@aol.com if you experience
ordering problems.
Greaseless high-vacuum with PTFE sealing ring for conical glass joints.
PTFE (polytetrafluorethylene) is equivalent to Teflon® from DuPont.
Download Technical Publication 1
(published in journal Fusion of the American Scientific Glassblowers Society ASGS).
Download Technical Publication 2
(Flyer with Tips and Tricks)

Prices of Glindemann PTFE Sealing Rings
End-user prices range from 20 € to 60 € per sales unit (unit = pack of 50 rings), dependent on distributor and order quantity.

1 sales unit = 1 pack of 50 PTFE-sealing rings on a plastic adapter (polypropylene) fitting the glass joint.
are sold by Dr. Dietmar Glindemann
Please contact Dr. Glindemann at dglinde@aol.com for sizes and a quotation.
,%20ohne%20Webadresse.jpg)
For hermetic handling of chemicals and environmental samples, syringe pump operation and cold storage.

For sale are gastight allglass-syringes, improved with an additional PTFE sealing ring in an additional groove; and replacement sealing rings.
Dr. Dietmar GLINDEMANN
Goettinger Bogen 15
D-06126 Halle
Germany
fx +49-(0)345-6871333.
E-mail dglinde@aol.com,
Internet : www.glindemann.net
Schlenk techniques with Glindemann-rings - Example uses of Glindemann-ring (PTFE sealing ring) for conical joint of Schlenk-flask with air- and moisture-sensitive chemicals, to exclude grease and improve cristallinization of large molecule analytes for spectrometry. Literature:
Chernichenko, K.
et al. Hydrogen activation by 2-boryl-N,N-dialkylanilines: a revision of
Piers’ ansa-aminoborane. Dalton Trans., 2012, 41, 9029-9032, DOI:
10.1039/C2DT30926B, see Electronic Supplementary Material file:
http://www.rsc.org/suppdata/dt/c2/c2dt30926b/c2dt30926b.pdf.
Chernichenko, K. et al. A frustrated-Lewis-pair approach to catalytic
reduction of alkynes to cis-alkenes. Nature Chemistry 5, 718–723 (2013),
doi:10.1038/nchem.1693, see Electronic Supplementary Material file:
http://www.nature.com/nchem/journal/v5/n8/extref/nchem.1693-s1.pdf.
Chernichenko, K. et al. Metal-Free sp2-C–H Borylation as a Common Reactivity
Pattern of Frustrated 2-Aminophenylboranes. J. Am. Chem. Soc., 2016, 138
(14), pp 4860–4868, DOI: 10.1021/jacs.6b00819, see Electronic Supplementary
Material file:
http://pubs.acs.org/doi/suppl/10.1021/jacs.6b00819/suppl_file/ja6b00819_si_001.pdf.
Wipf, P. (2014) Techniques for Handling Air- and Moisture-Sensitive
Compounds. See at
http://ccc.chem.pitt.edu/wipf/Web/Air_sensitive_techniques.pdf
.
Knudsen,
B. P., Chorkendorff, I., & Stephens, I. (2016). Synthesis of Platinum Rare
Earth Alloy Catalysts for Fuel Cells. Department of Physics, Technical
University of Denmark.
http://orbit.dtu.dk/files/127670440/Brian_Knudsen_PhD_Thesis_v1_reduced..pdf
Scott E. Denmark, Christopher R. Butler.
Vinylation with Inexpensive Silicon-Based Reagents: Preparation of
3-Vinyl-quinoline and 4-Vinylbenzophenone. Organic Syntheses, Vol. 86 (2009)
274-286.
Saleh et al. Chlorination of 1‑Carba-closo-dodecaborate and
1‑Ammonio-closo-dodecaborate Anions. Inorg. Chem. 2016, 55, 20, 10617-10627.
Schlenk flask greasefree tight by a combination of Glindemann-ring (PTFE) and PTFE-Valve. The thin PTFE ring becomes transparent under sealing pressure. PTFE (polytetrafluorethylene) is equivalent to Teflon® from DuPont.
Storage or freezing of air- and moisture-sensitive chemicals. Avoidance of contamination and leakage by leached-out joint-grease. Improved analytical crystallization for spectromety, undisturbed by grease contamination of analyzed chemical.
Short description of the main business of Dietmar GLINDEMANN (PTFE Sealing Ring for conical glass joints)
Laboratory work with chemical glass equipment and containers often demands hermetic and clean sealing of conical joints and stoppers. Joint grease is not solvent resistant and not clean. Commonly known PTFE-joint sleeves and PTFE-stoppers are expensive and not very tight.
Dietmar Glindemann solved these problems by inventing a fine PTFE-ring seal for ground tapered glass joints. The ring is inexpensive, gas-tight, solvent-tight, chemically inert, temperature resistant and prevents freezing of joints.
Inventor: Dietmar Glindemann (Technische Hochschule "Carl Schorlemmer" in Merseburg, Germany)
Production: Dietmar Glindemann.
Distribution: To see a list of distributors of GLINDEMANN® sealing rings and their catalogue numbers click on the "Business" part of this site.
Back to top
Short description of the Research and of other inventions of Dietmar GLINDEMANN
Biotechnology (Biocides, Food conservation, Phytochemicals)
Physicochemistry (separation, distillation, chromatography)
Trace analysis of volatile toxins, odorants containing heteroelements (sulfides, phosphine, organophosphines and other organoelement compounds)
Advanced gas chromatographic (GC) trapping techniques
GC-ICP interface construction
Odor research - chemical analysis of odorants, odor measurement by olfactometry, odor evaluation
DMSO (dimethyl sulfoxide) waste residues, DMSO reduction to dimethyl sulfide (DMS) and mitigation of sewage plant odor (City of Philadelphia).

Learn about why metals smell metallic (metallic odor, iron odor, copper odor, odor of money, smell of money... Link to the paper "The Two Odors of Iron..."
Artwork: D. Glindemann / Angew. Chem.
(click to enlarge)
About my domain www.phosphine.net (it links to www.glindemann.net)
I invented in 1993 an exceptionally sensitive and selective gas chromatographic method to detect traces of the gas Phosphine (Phosphane, PH3) in the environment. Using my sensitive detection method, I was the first scientist able to discover phosphine gas to be a global trace component of the earth’s atmosphere - it is everywhere. I produced extensive knowledge concerning the existence and formation mechanisms of phosphine everywhere in the earth’s natural and human made environment. I, and collaborators, discovered phosphine in waste gas, marsh gas, biogas, landfill gas, wetland sediment, soil, intestinal tract of mammals, faecal sludge, animal manure, landfill, technical metals, industrial waste, stone minerals, food and cosmic fall down (meteorites).
Keywords:
Glindemann (misspelled Glindeman Lindemann Lindeman)
Glass ground conical taper standard joint PTFE sealing ring seal, gasket, socket, sleeve, stopper, plug, leakage, vacuum, pressure, grease, jamming freezing, Schlenk techniques, Schlenk-flask, air sensitive chemicals, moisture sensitive chemicals,
Phosphine (CAS. 7803-51-2, PH3, Phosphane), Diphosphine (P2H4 , Diphosphane) toxic, environment, marsh gas, biogas, landfill gas, wetland sediment, air, food, organometallic, methyl mercury, dimethyl mercury (CAS 62-38-4, 593-74-8.), Organotin, tetramethyl tin (CAS 594-27-4), trimethyl tin (CAS 1066-45-1), Tributyltin chloride ( CAS 1461-22-9), octyl tin, phenyl tin, Tetraethyl lead (CAS 78-00-2), Tetramethyl lead (CAS 75-74-1)